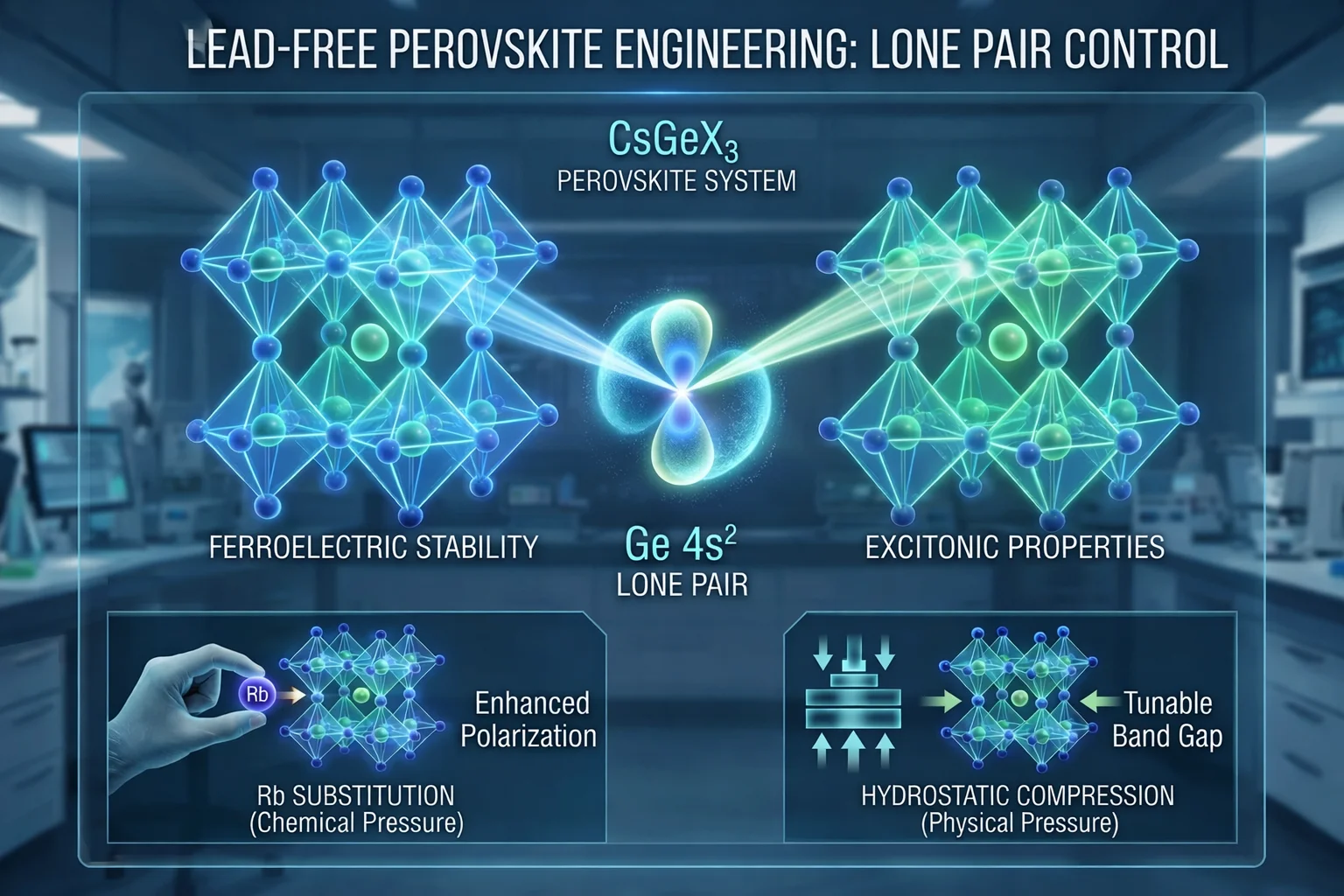
A groundbreaking study published in Scientific Reports on February 28, 2026, has established a unified microscopic framework for understanding and controlling the properties of lead-free halide perovskites, specifically the CsGeX3 system (where X = Cl, Br, I). Researchers Shahrbano Rahimi and S. Jalali-Asadabadi have identified that the stereochemically active Ge 4s² lone pair is the critical factor governing both ferroelectric stability and excitonic properties. This discovery addresses a long-standing challenge in materials science: achieving simultaneous control over optical absorption and ferroelectric stability in sustainable, non-toxic optoelectronic materials.
Lead-free halide perovskites are widely viewed as the future of sustainable technology, promising to replace toxic lead-based counterparts in solar cells and sensors. However, optimizing their performance has been difficult due to the complex interplay between their structural and electronic properties. By utilizing first-principles many-body calculations combined with polarization analysis and real-space charge mapping, the study reveals that electronic asymmetrydriven by the localization of the lone pairis more influential than lattice tetragonality in determining ferroelectric strength across the halide series.
The Role of Lone Pair Engineering
The core finding of the research is that lone pair localization serves as a transferable descriptor that connects charge redistribution, dielectric response, and excitonic confinement. The researchers demonstrated that the Ge 4s² lone pair does not merely sit passively within the crystal structure; it actively dictates the material's electronic landscape. This insight allows scientists to predict how changes in chemical composition will affect the material's ability to store electric charge and interact with light, effectively establishing "lone pair engineering" as a general strategy for designing multifunctional materials.
Tuning Properties via Rb Substitution and Compression
The study explicitly details two methods for manipulating these materials to achieve desired outcomes. First, the researchers found that partial substitution with Rubidium (Rb) acts as a form of "chemical pressure." This substitution enhances polarization and stabilizes the ferroelectric state while crucially preserving visible light absorption and avoiding the creation of detrimental mid-gap states. This suggests a pathway to create stable, high-performance materials for optical devices.
Second, the team explored hydrostatic compression as a reversible method to tune the material's properties. Unlike chemical substitution, which permanently alters the composition, physical compression allows for the dynamic adjustment of exciton binding energies and band gap energies. This dual approachchemical pressure for stability and physical pressure for tunabilityprovides a comprehensive toolkit for developing next-generation optoelectronic devices.
My Take
This research represents a pivotal shift in how we approach materials design for green technology. By moving the focus from macroscopic lattice structures to microscopic electronic asymmetry (specifically the Ge 4s² lone pair), the authors have provided a roadmap for overcoming the stability issues that have plagued lead-free perovskites. For the industry, this means the potential for high-efficiency, non-toxic solar panels and photosensors is closer to reality. The distinction between using Rb substitution for permanent stability and hydrostatic compression for reversible tuning offers versatile options for device engineers looking to tailor materials for specific environmental conditions.