Table of Contents
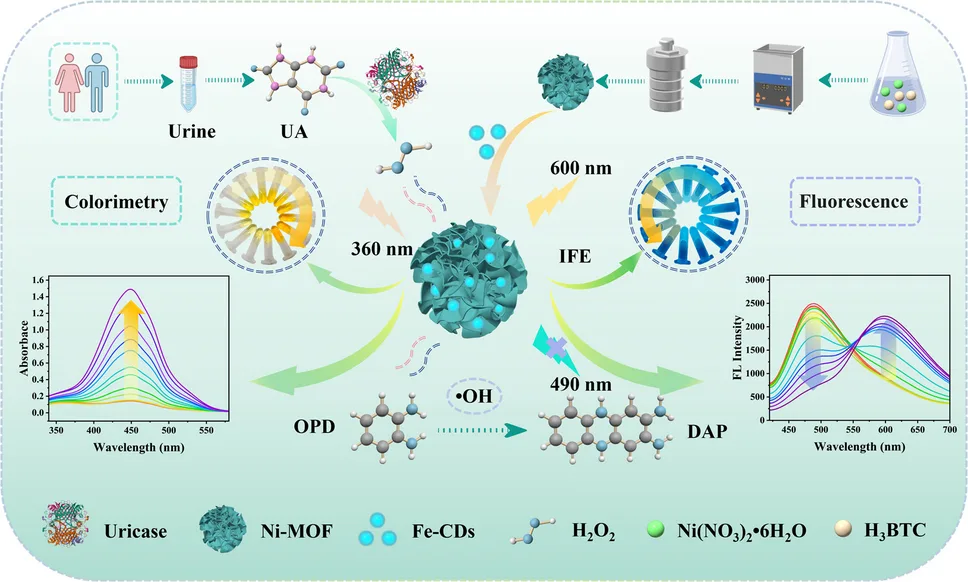
Ni-MOF@Fe-CDs nanozyme probe revolutionizes uric acid detection with dual colorimetric and ratiometric fluorescence signals, achieving a detection limit of 0.072 μM. Led by Chunyan Yan, Bowen Yu, and Ping Qiu, this innovation loads iron-carbon dots onto Ni-MOF nanoflowers for superior peroxidase-like activity.
Chemists and bioanalysts targeting precise biomarkers in clinical samples gain a tool for rapid, visual uric acid quantification, enabling early gout diagnosis and metabolic disorder monitoring.
The probe generates hydroxyl radicals (•OH) from H2O2, confirmed by electron paramagnetic resonance, with steady-state kinetics showing Km of 1.18 mM and Vmax of 9.82 × 10 - 8 M•s - 1. This outperforms traditional enzymes in stability and cost.
Nanozyme Design and Catalytic Mechanism
Fe-CDs integrate into Ni-MOF petals, enhancing electron transfer for efficient H2O2 decomposition. The peroxidase-mimicking activity oxidizes o-phenylenediamine (OPD) in the presence of H2O2 produced from uric acid via uricase.
2,3-Diaminophenazine (DAP) from OPD oxidation quenches Ni-MOF@Fe-CDs fluorescence at 430 nm via inner filter effect (IFE), dominating 85.2% of quenching efficiency. This ratiometric shift (FI565/FI430) ensures self-calibrating, interference-resistant detection.
For researchers in analytical chemistry, this dual-function platform verifies results across modes, reducing false positives in complex matrices like urine where uric acid levels signal health risks such as gout or kidney injury.
Performance in Real Samples
In human urine, recoveries range 92.2 - 107.6% with RSD <2.9%, matching clinical standards. The visual colorimetric mode suits point-of-care testing, while fluorescence offers lab-grade precision.
Compared to single-mode sensors, dual outputs provide mutual validation, critical for biomarkers like uric acid linked to multiple sclerosis disability and hypouricemia-related kidney issues.
Broader Context and Significance
Uric acid monitoring matters in metabolism; elevated levels (>420 μM in men, >360 μM in women) precede gout, while lows indicate hereditary risks. Nanozyme tech like Ni-MOF@Fe-CDs scales production cheaply versus natural enzymes.
This advances from prior MOF sensors, such as Fe-Ni-MOF/RGO (LOD 0.021 μM electrochemical) or Fe3Ni-MOF-NH2 (24 nM ratiometric), by combining catalysis and fluorescence in one probe.
My Take
With a 0.072 μM LOD and real-sample recoveries over 92%, Ni-MOF@Fe-CDs positions as a frontrunner for portable diagnostics, potentially slashing gout misdiagnosis rates that affect 4% globally. Its IFE-dominant quenching (85.2%) signals robust anti-interference, forecasting integration into wearables for continuous uric acid tracking amid rising metabolic diseases.
Frequently Asked Questions
What is the detection limit of Ni-MOF@Fe-CDs for uric acid? The limit is 0.072 μM in dual-mode detection.
How does it perform in urine samples? Recoveries are 92.2 - 107.6% with RSD <2.9%.
What makes it dual-signal? Colorimetric via OPD oxidation and ratiometric fluorescence via DAP quenching of Fe-CDs emission.