Table of Contents
Researchers have developed a highly efficient method for electrochemical propylene epoxidation from seawater, achieving a near-perfect 99.0% Faradaic efficiency. By incorporating lithium into cobalt oxide catalysts, the scientific team successfully synthesized propylene oxide, paving the way for sustainable chemical production using renewable electricity and waste materials. The findings, published in Nature Communications on March 14, 2026, represent a major leap forward in green chemistry.
This breakthrough is crucial for chemical engineers, materials scientists, and industrial manufacturers seeking sustainable alternatives to traditional petrochemical processes. By utilizing abundant seawater and recycled lithium, industries can drastically reduce the environmental footprint and energy costs associated with commercial chemical synthesis. This approach enables a direct transition away from fossil-fuel-dependent manufacturing toward a circular, electrified economy.
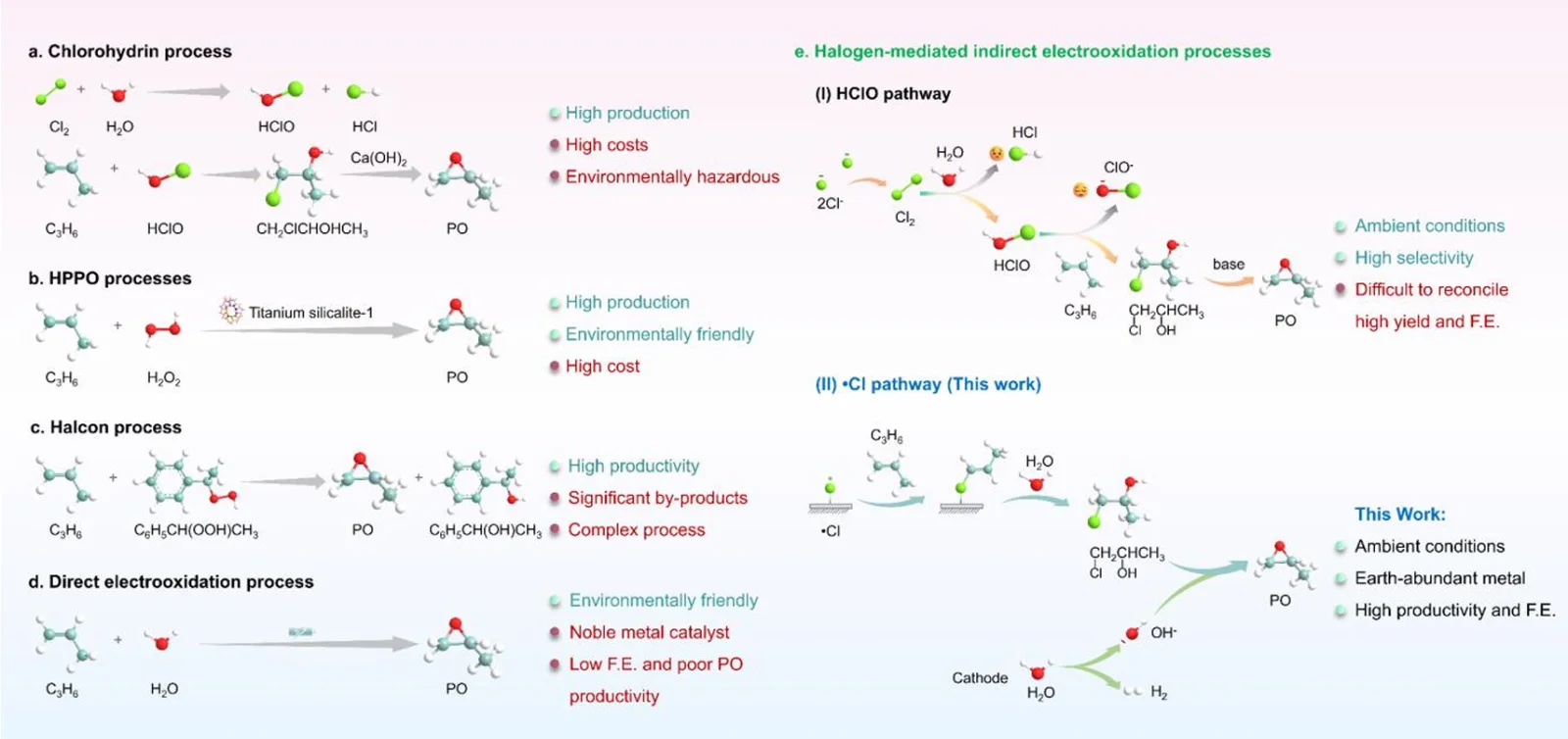
Propylene oxide (PO) is a vital commercial chemical used globally in the production of plastics, polyurethanes, and industrial solvents. Traditional synthesis methods are notoriously energy-intensive and environmentally taxing, often generating significant toxic byproducts. Electrocatalytic chlorine-mediated propylene oxidation using seawater has recently emerged as a promising green alternative. However, previous methods suffered from the inefficient utilization of active chlorine species, which ultimately led to compromised yields and low Faradaic efficiency.
The Lithium-Doped Co3O4 Breakthrough
The research team, which includes Ming Cheng, Xiaoxian Sun, Peng Zhang, Xiao Liu, and Jinlong Gong, solved this critical efficiency problem by doping a standard cobalt oxide (Co3O4) catalyst with lithium (Li). This precise atomic modification achieved a remarkable PO yield of 20.6 mol/m2/h in simulated seawater. Furthermore, the newly engineered catalyst maintained exceptional operational stability for over 100 hours without degradation.
Crucially, the catalyst demonstrated highly competitive performance even when scaled to industrial parameters. Under an industrial-level current density of 0.5 A cm-2, the system achieved an impressive PO yield of 78.9 mol/m2/h. This level of performance rivals or exceeds most currently reported noble metal catalysts, making the lithium-doped Co3O4 highly viable for large-scale commercial deployment in chemical plants.
Mechanistic investigations revealed exactly why this specific lithium doping is so effective. The addition of lithium fundamentally transforms the adsorption configuration of chloride ions (Cl - ) on the Co3O4 surface. Instead of forming a standard linear Co-Cl structure, the ions arrange into a more thermodynamically favorable triangular Li-Cl-O configuration.
This structural shift directly enhances the generation of chlorine radicals (•Cl) during the electrochemical process. Consequently, it redirects the entire propylene oxidation process away from the conventional, less efficient HClO pathway. By forcing the reaction down the highly efficient •Cl route, the system maximizes the conversion rate and minimizes wasted energy.
My Take
The integration of waste lithium-ion batteries as a potential source for the lithium dopant, combined with abundant seawater and renewable electricity, creates a brilliant closed-loop approach to chemical manufacturing. As global industries face mounting legislative pressure to decarbonize, this specific •Cl pathway optimization proves that non-noble metal catalysts can outperform expensive traditional alternatives when engineered at the atomic level. The ability to sustain a yield of 78.9 mol/m2/h at industrial current densities indicates that this technology is not just a laboratory curiosity, but a commercially ready solution that could disrupt the traditional propylene oxide market.
Frequently Asked Questions
What is propylene oxide used for?
Propylene oxide is a highly versatile commercial chemical primarily used to manufacture polyurethanes, plastics, and various industrial solvents.
Why use seawater for this electrochemical process?
Seawater provides an abundant, natural source of chloride ions (Cl - ), which are essential for the chlorine-mediated oxidation process, eliminating the need for purified chemical feedstocks.
How does lithium improve the catalyst's performance?
Lithium changes the adsorption configuration of chloride ions on the catalyst surface to a triangular Li-Cl-O structure, which boosts the generation of chlorine radicals and significantly increases the reaction's efficiency.