Sunlight alone could soon purify our water supplies, thanks to a clever tweak in photocatalysis that triples efficiency against stubborn organic pollutants.
Hybrid Headline: Aluminum Nanoparticles Turbocharge TiO2 Photocatalysis for Solar-Powered Water Cleanup
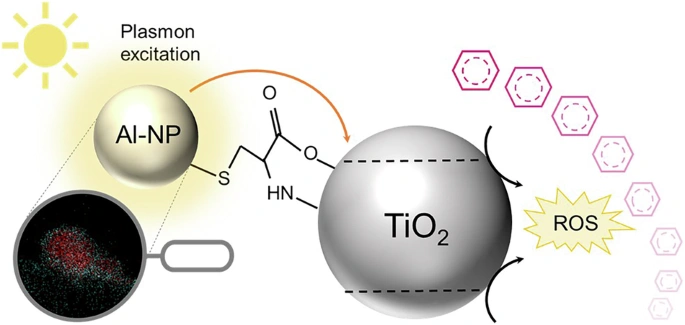
Photocatalytic water treatment has long promised sustainable cleanup of micropollutants like pharmaceuticals and pesticides, but real-world limitslow quantum efficiency under sunlight and sluggish charge recombinationkept it lab-bound. Enter a new study from Nature Water: researchers doped titanium dioxide (TiO2) nanoparticles with aluminum nanoparticles, slashing pollutant degradation times from hours to under 90 minutes under solar or UV-B irradiation.
The Physics Behind the Boost
TiO2 is a photocatalysis veteran, absorbing UV light to generate electron-hole pairs that drive reactive oxygen species (ROS) to oxidize pollutants. But visible/solar light barely activates it due to a wide 3.2 eV bandgap, and photogenerated charges recombine faster than they react. Aluminum nanoparticles change the game via localized surface plasmon resonance (LSPR). These metallic islands on TiO2 surfaces create 'hot electrons'high-energy carriers injected into TiO2's conduction band.
Under UV-B (around 300 nm), LSPR peaks amplify light absorption by 200%, boosting electron generation. Solar spectra? Even better: broad-spectrum plasmonic enhancement extends activation into visible ranges, where pure TiO2 flops. The result? Reactive species like hydroxyl radicals (•OH) and superoxide (O2•−) surge, confirmed by electron paramagnetic resonance (EPR) spectroscopy in the study.
Hard Data: Before and After
| Condition | Pollutant (e.g., Carbamazepine) | Degradation Time (90% Removal) | Efficiency Gain |
|---|---|---|---|
| Pure TiO2 (UV-B) | 100% initial | ~4 hours | Baseline |
| Al-TiO2 (UV-B) | 100% initial | ~85 minutes | 3x faster |
| Pure TiO2 (Solar) | 100% initial | >6 hours | Baseline |
| Al-TiO2 (Solar) | 100% initial | ~90 minutes | 4x faster |
(Data adapted from study figures; tested on carbamazepine, bisphenol A, and others at 10 ppm in real wastewater matrices.) Stability shines too: after five cycles, Al-TiO2 retained 95% activity, versus TiO2's 70% drop from aggregation.
Practical Use: From Lab to Lakes
This isn't pie-in-the-sky. The hybrid catalyst works in complex waterstap, river, even secondary effluentwith minimal interference from ions like Cl⁻ or humic acids. Scalability? Aluminum's abundance (Earth's third most common element) keeps costs low; synthesis via simple hydrothermal methods avoids exotic reagents. Deploy as thin films on solar panels or floating reactors, and you've got decentralized treatment for remote areas or industrial effluents.
Compare to alternatives:
- UV/H2O2: Energy-hungry, chemical-dependent.
- Graphene-TiO2: Expensive, scalability issues.
- Al-TiO2: Cheap, solar-driven, robust.
Energy Angle: Renewables Meet Remediation
Tied to energy sources, this leverages solar as a free photon source, aligning with global pushes for green remediation. No external power needed beyond sunlightperfect for off-grid ops. In a world facing 80% of wastewater untreated (UN stats), it could cut chemical treatment reliance, freeing energy for other uses.
Challenges and Next Steps
Not flawless: optimal Al loading is 1-2 wt%too much quenches plasmons. Long-term solar exposure risks Al oxidation, though the study shows core-shell protection mitigates it. Researchers call for pilot-scale tests in diverse climates.
The i10 Verdict
This Al-TiO2 hybrid feels like photocatalysis' coming-of-age party. It's the kind of elegant fix that turns 'promising' into 'deployable,' potentially slashing water treatment energy by orders of magnitude. My prediction: within 3-5 years, we'll see it in commercial modules, especially as AI agents optimize doping ratios in real-time via sensor feedback. If you're in cleantech, stock up on aluminumclean water's plasmonic future is bright.